 Micelles occur when a spherical fatty acids structure is formed with a hydrophobic core and hydrophillic outer shell. The hydrophobic areas of amphipathic molecules cluster together to avoid the ordered "cage" of water molecules that would surround them and orient the hydrophillic ends as a shield-like outer structure that interacts amicably with the polar water molecules. 
Hydrophobic interactions can also be seen in the clustering of amphipathic/amphiphillic molecules such as phospholipids into bilayers and micelles. The favorable release of water molecules from nonpolar surfaces is responsible for phenomenon of the hydrophobic effect. The water molecules that initially "caged" the nonpolar molecules are released from the nonpolar molecules' surfaces, creating an increase in entropy in the surrounding. Therefore, it is favorable for the nonpolar molecules to associate without the interference of water. 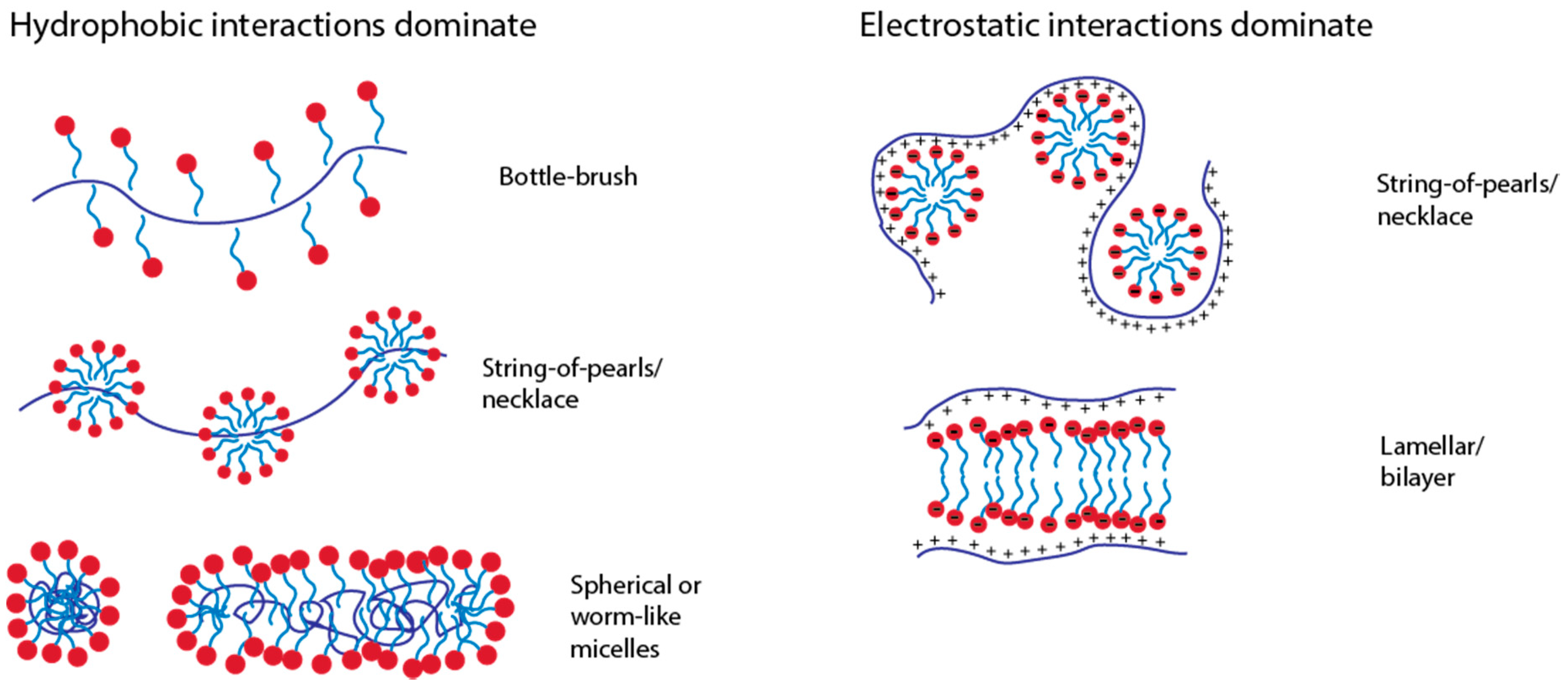 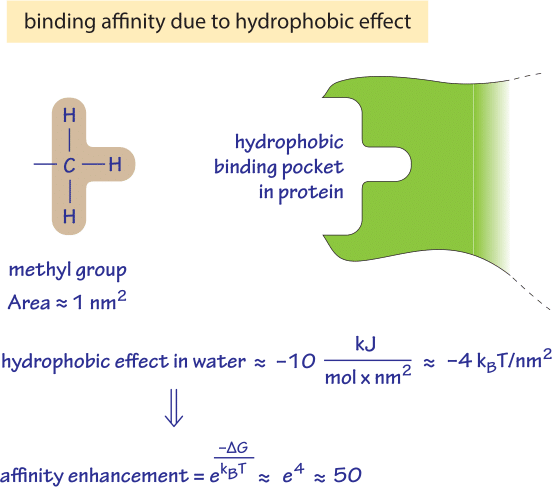
Hydrophobic effect entropy plus#However, the tendency of nonpolar molecules to associate with one another will draw the nonpolar molecules together, forming a nonpolar aggregate.īased on the second law of thermodynamics, the total entropy of the system plus its surrounding must always be increasing. When nonpolar molecules are introduced to the water molecules, the water molecules will initially surround the nonpolar molecules, forming a "cages" around the molecules. The interactions between nonpolar molecules and water molecules are not as favorable as interactions amongst just the water molecules, due to the inability of nonpolar molecules to form hydrogen bonding or electrostatic interactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
